
Once TPO is banned, where will UV LED curing go?
Recently, the European Chemicals Agency (ECHA) officially announced that Diphenyl(2,4,6-trimethylbenzoyl)phosphine oxide will be included in the 29th batch of Substances of Very High Concern (SVHC) candidate list. So far, the number of SVHC candidate list substances has increased to 235.
For chemicals on the list, companies are responsible for risk management of the chemicals and providing information to customers and consumers about the safe use of these chemicals. These substances may be included in the authorization list in the future. If a substance is on the list, its use will be banned unless companies apply for authorization from the European Commission to continue their use.
The specific information of the new substances is as follows:
Substance: Diphenyl(2,4,6-trimethylbenzoyl)phosphine oxide
Synonym: Photoinitiator TPO;SINOCURE TPO
EC number: 278-355-8
CAS No.:75980-60-8
Reason for inclusion: Reproductive toxicity (Article 57(c))
Common uses: in inks and toners, coating products, photochemical polymers, adhesives and sealants and fillers, plaster modeling clay, etc.
-
01.The development path of China’s Photocuring
Photocuring refers to the curing process of monomer, oligomer or polymer matrix under light induction, and is generally used in the film-forming process. The technology has the characteristics of high efficiency, wide adaptability, economy, energy saving and environmental protection.
Photocuring is mainly divided into traditional mercury lamp curing and emerging UV LED curing. Because traditional mercury lamps will cause serious pollution to the environment without proper treatment after use, UV LED curing has many advantages such as more energy saving, can be switched on and off at any time, and is smaller in size. It is gradually replacing traditional mercury lamp curing and becoming the light source. Mainstream of curing equipment.
The proportion of photoinitiators in photocuring formulas is not high, usually around 2%-5%, but it plays an important role. Because of the photocuring reaction, the photoinitiator needs to absorb ultraviolet light to generate free radicals, thereby initiating the polymerization reaction and finally solidifying the product.
Traditional photoinitiators, such as 1173, 184, etc., have their maximum absorption wavelength at the UVC short wavelength, so traditional mercury lamp curing is more suitable.
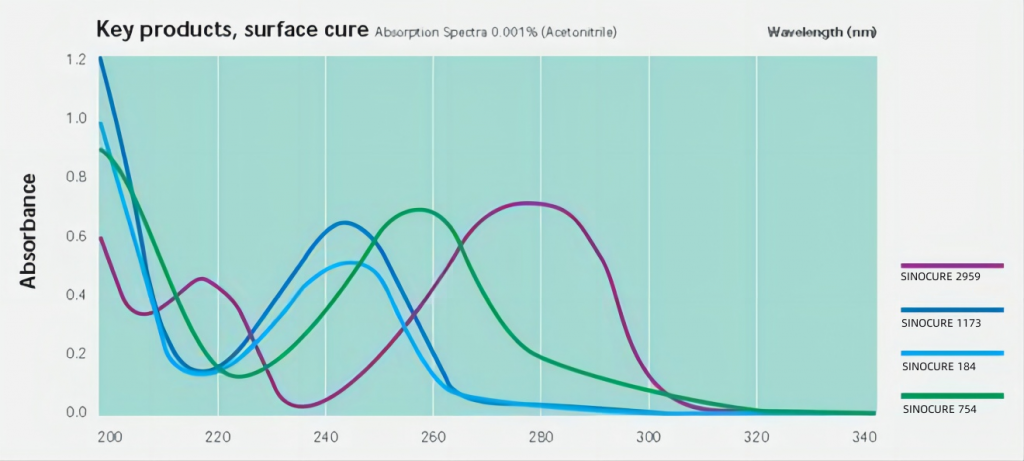
UV LEDs are mainly concentrated in a few wavelength bands such as 365nm, 385nm, 395nm and 405nm. Phosphine oxide photoinitiators have relatively strong absorption in these bands, so they are widely used in UV LED applications.
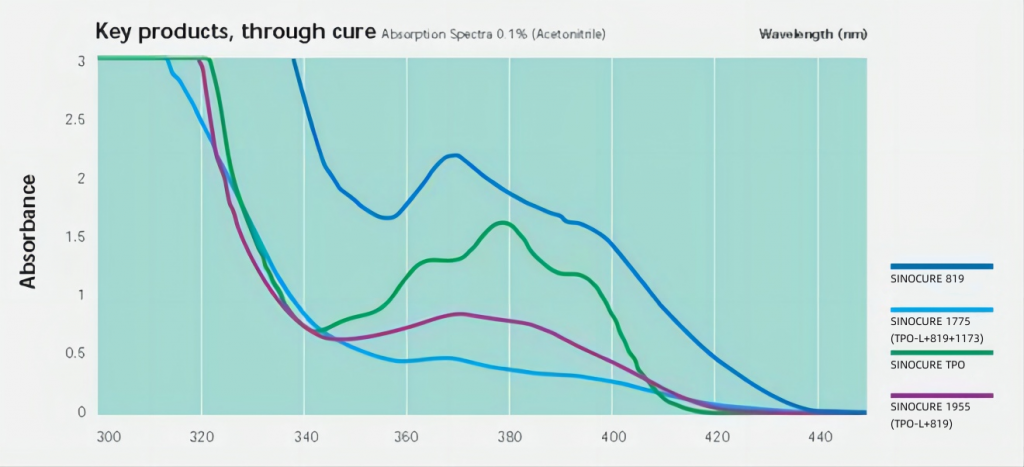
One of the most representative photoinitiators is TPO. It not only has high inducing efficiency, non-yellowing, but is also relatively affordable. However, in the past few years, due to the vigorous rise of UV LED curing, the global supply of TPO has been tight and it is difficult to find a single product. In recent years, due to the continuous expansion of domestic mainstream photoinitiator manufacturers and the successive entry of new manufacturers, TPO The tight supply situation has been greatly alleviated, and prices have returned to normal levels. The normal supply of TPO has also greatly promoted the development of UV LED.
-
02.Toxicity classification and restricted use of TPO
Photoinitiators are usually small-molecule organic compounds. When illumination is incomplete, these photoinitiator molecules will remain in the cured product, forming potential migration substances. In addition, in most cases, the process of generating free radicals from photoinitiators is through cleavage. These free radicals may form some smaller molecular weight compounds after final quenching. The products of these small molecules not only cause migration problems, but may also produce some toxic substances.
With the widespread use of photoinitiator TPO, its supervision is also strengthening. According to the EU’s CLP (Classification, Labeling and Packaging) regulations, TPO was initially classified as a Category 2 (H361) reproductive toxicant, that is, “Suspected human reproductive toxicant”.
In June 2020, the Nordic country Sweden proposed to modify its classification to 1B (H360DF) and add it as a skin irritating substance (H317). This is based on evidence from numerous animal experiments. (1B refers to “Presumed human reproductive toxicant”
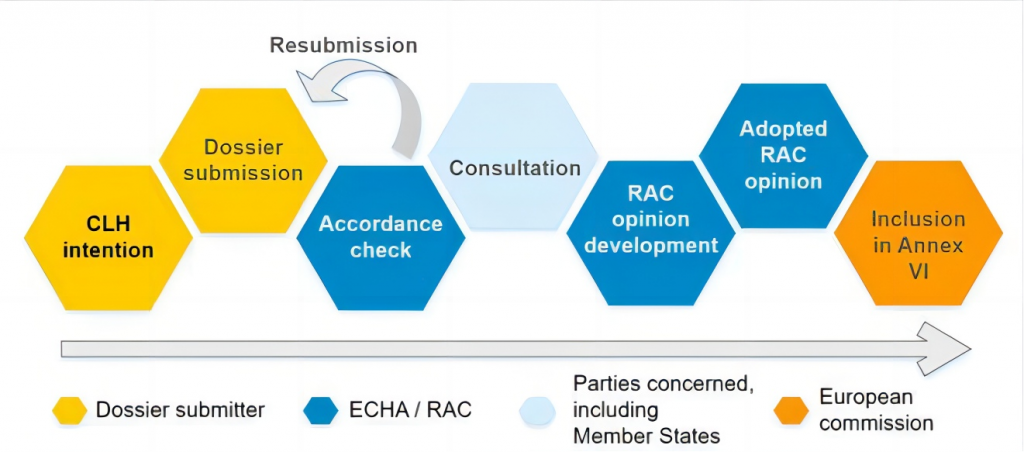
EU harmonized classification and labeling (CLH) process
In the fall of 2021, the European Union’s Risk Assessment Committee (RAC) agreed to update the classification of TPOs. Once approved by the European Commission, the classification will be added to Annex VI of the EU CLP Regulation through ATP and become legally binding.
In January 2023, Sweden announced a notice of intention to propose that TPO be included in the SVHC (Substances of Very High Concern) list. Comments on the proposal ended on April 3, 2023.
So far, TPO has been included in the 29th batch of Substances of Very High Concern (SVHC) candidate list.
-
03.Alternative options for photoinitiator TPO
Among phosphine oxide photoinitiators with good absorption capacity in the UVA band, in addition to TPO, there are two other commonly used photoinitiators, SINOCURE TPO-L and SINOCURE 819 (BAPO).
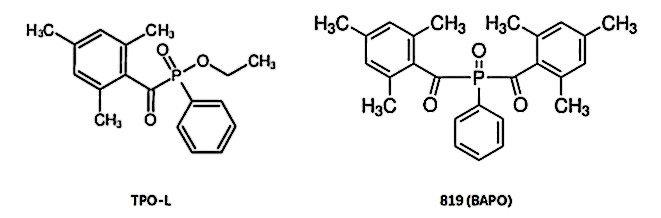
TPO-L has a similar structure to TPO, but is less toxic because one of the benzene rings in the molecule is replaced with an ethoxy group. But the disadvantage is that the priming efficiency of TPO-L is much lower than that of TPO.
Another phosphine oxide photoinitiator is 819 (BABO), which can be regarded as a benzene ring in TPO being replaced by a 2,4,6-trimethylbenzoyl group, that is, it has two 2,4,6 -Trimethylbenzoyl group. The initiating efficiency of 819 is higher than that of TPO, but it has a serious yellowing problem, which cannot be used in situations where color is required.
In other words, TPO-L and 819 can only replace TPO in some application scenarios, but they cannot completely replace it.
The full name of SINOCURE 2425 is (2,4,6-trimethylbenzoyl)bis(p-tolyl)phosphine oxide, CAS 270586-78-2. From an intuitive structural point of view, SINOCURE 2425 introduces a methyl group on each of the two benzene rings on the basis of TPO, which greatly reduces the biological toxicity of TPO.
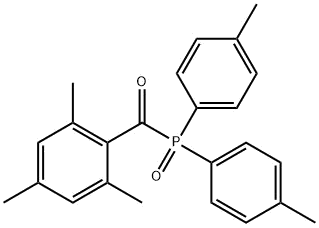
After experiments, it was found that the initiation efficiency of SINOCURE 2425 is even slightly better than that of TPO, while it does not yellow and has lower migration.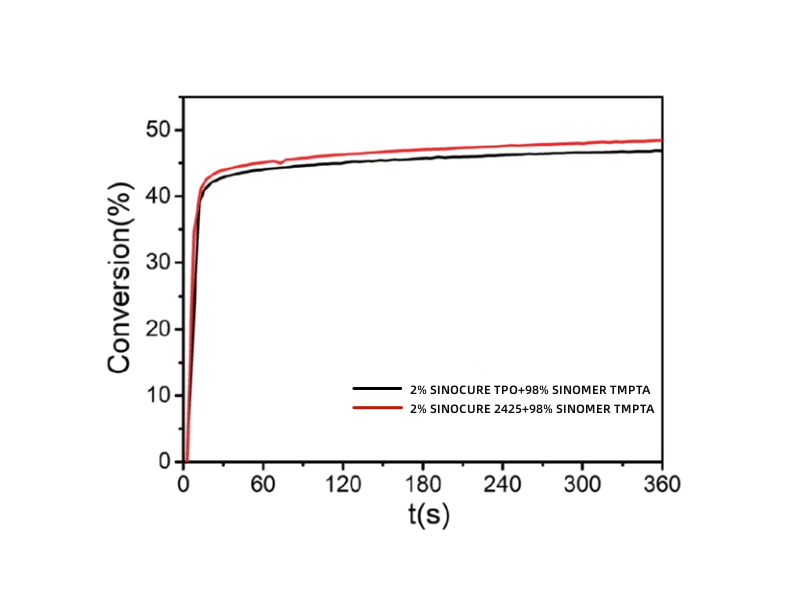
Double bond conversion rate curve of TMPTA triggered by SINOCURE 2425 and SINOCURE TPO
At present, SINOCURE 2425 has achieved mass production and has obtained the EU’s “Regulation, Evaluation, Authorization and Restriction of Chemicals” (“REACH”) registration certificate, and can be sold to European regions with the strictest chemical controls.
